Amyloidosis and Cancer Theranostics Program
Program Overview
Amyloidosis is a relatively rare protein-folding disorder characterized by the formation of well-structured protein aggregates, fibrils, that deposit in organs and tissues. In addition to fibrils, amyloid deposits contain extracellular matrix proteins, such as collagen and heparan sulfate proteoglycans, and other proteins sequestered from the blood. The unrelenting accumulation of amyloid often leads to organ dysfunction and severe morbidity. Systemic amyloidosis can affect any organ or tissue, but the heart, kidneys, pancreas, liver, spleen, and nerves constitute the major sites of deposition in patients with familial or sporadic forms of systemic amyloid disease.
The major types of systemic amyloidosis are associated with the deposition of immunoglobulin light chains (AL amyloidosis), wild type or mutant transthyretin (ATTRwt or ATTRv amyloidosis), and leukocyte chemotactic factor 2 (ALECT2 amyloidosis). Altogether, there are approximately 20 diverse types of systemic amyloidosis, each one characterized by the type of protein that forms the fibrils.
The treatment of amyloidosis has seen remarkable advances in the development of new reagents over recent years, including gene silencers and protein stabilizers for patients with ATTR amyloidosis and immunotherapy for patients with AL amyloidosis. These novel drugs have led to enhanced diagnosis and much-improved outcomes for these patients.
However, there are currently no FDA approved methods for clearing amyloid from organs and tissues, which could be curative, and no approved method for diagnosing the whole-body distribution of amyloid. To address these clinical unmet needs, the ACTP has, over the last decade, developed amyloid-reactive antibodies and peptide agents (“biologicals”) for the treatment and detection of amyloid deposits.
With the support of the National Heart, Lung, and Blood Institute at the NIH, a peptide, designated p5+14, was developed that could bind all forms of amyloid. When labeled with a radioactive molecule, the peptide was capable of imaging all visceral amyloid deposits in a mouse model of systemic amyloidosis by using PET/CT or SPECT/CT imaging. The ACTP has developed this peptide as an imaging agent for detecting systemic amyloid of any type in patients. With the support of the University of Tennessee Medical Center (Depts. of Nuclear Medicine and Cardiology, and the Cancer Institute) this peptide was studied in a first-in-human PET/CT imaging study to assess safety and the binding to amyloid in the heart and other organs.
The ACTP is now working to develop additional imaging radiotracers and therapeutic agents for removing amyloid from organs. To this end, modified pan-amyloid reactive antibodies and cells with enhanced clearing capabilities are under investigation. Additionally, new studies in oncology and the development of potentially therapeutic oncolytic viruses are underway.
The ACTP is dedicated to studying all aspects of amyloidosis, particularly AL amyloid, that impact patient outcomes and performing translational research to develop novel ways of improving these outcomes.
Research Team
- Jonathan Wall, PhD, Distinguished Professor, Program Director
https://orcid.org/0000-0002-5516-8578 - Stephen Kennel, PhD, Professor
- Emily Martin, PhD, Associate Professor, clinical research, risk assessment assay development
https://orcid.org/0000-0003-4993-5782 - Manasi Balachandran, PhD, Assistant Professor, Immunology and Assay development
https://orcid.org/0000-0002-9804-7426 - Joseph Jackson, PhD, Assistant Professor, Immunology, virology, and oncology
https://orcid.org/0000-0003-1572-6907 - Trevor Hancock, PhD, Immunology, virology, bioassay development
ttps://orcid.org/0000-0002-3353-6042 - Angela Williams, MS, Protein-peptide assays and development
- Sallie Macy, BS, MLT, ASCP, Immunohistochemistry
- Craig Wooliver, MLT, Histology and microscopy
- Tina Richey, MS, Preclinical animal models and compliance
- Steve Foster, MS, Cell biology and hybridomas
- Alan Stuckey, BA, CNMT, Preclinical animal imaging
- Marina Vlasyuk, MS, Preclinical oncology assay development

(Back): Steve Kennel, Steve Foster, Jon Wall, Craig Wooliver, Trevor Hancock. (Front): Tina Richey, Joseph Jackson, Emily Martin, Sallie Macy, Manasi Balachandran, Alan Stuckey, Angela Williams.
The Amyloidosis and Cancer Theranostics Program is developing novel biological therapeutic and diagnostic agents for patients with amyloidosis or cancer.
Clinical Studies
Amyloid Imaging for diagnosis and monitoring disease and therapy
The ACTP has been investigating novel ways to image amyloid using clinical PET/CT and SPECT/CT imaging techniques. The goal is to develop a clinical agent, or panel of agents, that can be used for detecting all types of amyloid throughout the body. We have designed and are testing agents that can be used in various clinical settings, from academic medical centers to cardiologist clinics. Our strategy relies on the use of the amyloid-binding peptide p5+14, labeled with various forms of radioactivity- principally iodine-124 for PET/CT imaging and technetium-99m for SPECT/CT studies.
In 2018, with support from the translational research SMARTT program offered by the NHLBI/NIH (now the Catalyze program) we were able to generate all the data required to submit an Investigational New Drug (IND) application to the FDA for a study to examine the safety and efficacy of the imaging agent in patients with amyloidosis and a small group of healthy subjects. This study, called AMY1001, was funded by generous donations to the ACTP gift fund and patients from all over the US travelled to Knoxville to participate.
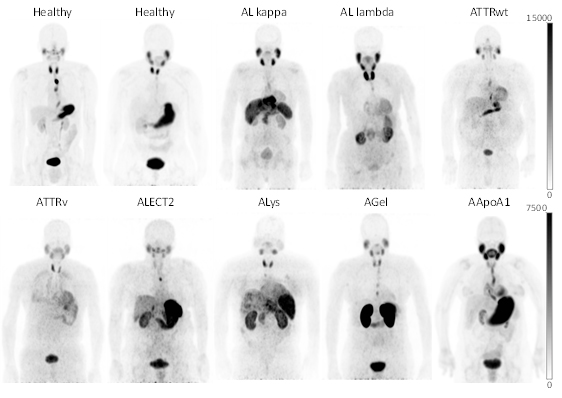
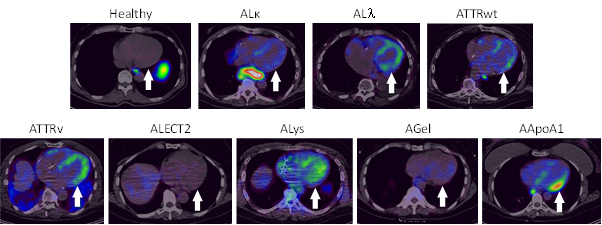
In late 2018 and early 2019, the first 3 amyloid patients underwent PET/CT imaging with increasing doses of 124I p5+14, now designated iodine-124-evuzamitide (but also called 124I-AT-01 and 124I-p5+14). These patients were carefully evaluated to ensure that the radioactive dose was within accepted limits and would not harm the patient. Over the following two years, 47 more patients with seven different amyloid types, two asymptomatic TTRv patients, and five healthy volunteers were imaged. The images we obtained (see below) show clear uptake of the tracer in systemic organs known to contain amyloid, including the heart. Based on the data generated in this study, both the European Medicines Agency (EMA) and the US Food & Drug Administration (FDA) have granted orphan drug designation to iodine-124-evuzamitide for the diagnosis of AL and ATTR amyloidosis (In the US for AL: DRU-202208884, and for ATTR: DRU-2022-8915. In the European Union, OD for AL: 0000096114. for ATTR: 0000096686).
Study ID: AMY1001: 124I-p5+14 Injection as a PET/CT Imaging Agent for Systemic Amyloidosis (IND 132282). Opened November 2018- Closed to enrollment August 2021 |
This was a pilot, first-in-human open-label Exploratory IND Phase I imaging and biodistribution study performed at the University of Tennessee Medical Center (UTMC). Patients were recruited from across Tennessee and the US. It was designed to determine the radiation dosimetry and capability of the amyloid-reactive, synthetic peptide, p5+14 (also then known as APi1832), radiolabeled with iodine-124 (I-124 or 124I) to image amyloid in organs or tissues of patients with any type of cardiac or other systemic amyloid deposits. |
Comparison of iodine-124-evuzamitide uptake in organs of healthy volunteers and patients with diverse types of systemic amyloidosis. From "Clinical Confirmation of Pan-Amyloid Reactivity of Radioiodinated Peptide 124I-p5+14 (AT-01) in Patients with Diverse Types of Systemic Amyloidosis Demonstrated by PET/CT Imaging". Pharmaceuticals 2023, 16, 629. https://doi.org/10.3390/ph16040629
Comparison of iodine-124-evuzamitide uptake in the heart of healthy volunteers and patients with diverse types of systemic amyloidosis. Clinical Confirmation of Pan-Amyloid Reactivity of Radioiodinated Peptide 124I-p5+14 (AT-01) in Patients with Diverse Types of Systemic Amyloidosis Demonstrated by PET/CT Imaging. Pharmaceuticals 2023, 16, 629. https://doi.org/10.3390/ph16040629
|
We continued to use this imaging agent to look at changes in amyloid load over time and recently completed a repeat imaging study, called UTMC-AT01-163117, in patients with AL and ATTR-associated amyloidosis.
Iodine-124-evuzamitide is ready for its Phase 3 evaluation and is currently being used by other clinician-researchers around the US, at: the Brigham and Women’s Hospital in Boston; Columbia University Irving Medical Center in New York; and the Oregon Health and Science University in Portland.
Study ID: UTMC-AT01-163117: Repeat PET/CT imaging of patients with systemic amyloidosis using amyloid reactive peptide 124I-AT-01 (124I-p5+14, Iodine-124I-evuzamitide) to measure changes in organ-specific amyloid load (IND# 163117). Opened December 2022 - Closed to enrollment July 2022 |
This was a single center, open label, prospective study using 124I-AT-01 PET/CT imaging to evaluate changes in amyloid load in up to 20 patients with systemic amyloidosis who had previously undergone 124I-AT-01 imaging at UTMC. As part of this study, in addition to PET/CT imaging, patients underwent a transthoracic echocardiographic examination. The previously acquired 124I-AT-01 PET/CT images will be used as baseline values to determine quantitative changes in uptake of the radiotracer in the heart, liver, spleen, and kidney (and other organs deemed positive following visual inspection of the baseline and repeat imaging) as a measure of changes amyloid load. |
A new version of the imaging agent has been developed and tested in the laboratory and is now ready for first-in-human testing at UTMC. This study called, UTGSM-164388-101, will look at the safety and biodistribution of technetium-99m-labeled peptide using planar and SPECT/CT imaging.
Study ID: UTGSM-164388-101: A Phase 1 Pilot Study to Investigate the Dosimetry and Biodistribution of a Single Intravenous Administration of 99mTc-p5+14 using SPECT/CT Imaging and Planar Gamma Scintigraphy in Healthy Volunteers and Patients with AL or ATTR Systemic Amyloidosis (IND# 164388) - Opened July 2023 |
This study will investigate 99mTc-p5+14, an amyloid-reactive synthetic peptide, p5+14, radiolabeled with technetium-99m, as a radiotracer for planar gamma scintigraphy (PGS), single photon emission computed tomography (SPECT) or SPECT with x-ray computed tomography (SPECT/CT) for the diagnosis of systemic amyloidosis, notably with cardiac involvement. Based on nonclinical data and clinical data of 124I-p5+14 from Study AMY1001, peptide p5+14 binds many types of human amyloid and is rapidly cleared from the blood. When radiolabeled with 99mTc, p5+14 may enable visualization of amyloid laden tissues and organs by SPECT/CT imaging or PGS. Thus, this study has been designed to evaluate the dosimetry and biodistribution of 99mTc-p5+14 in patients with systemic AL or ATTR amyloidosis compared with the biodistribution in healthy volunteers. |
Screening for amyloid deposits in tissue biopsies during routine surgery.
Certain conditions may precede the development of systemic amyloidosis by nearly a decade. These could include lumbar spinal stenosis, bilateral carpal tunnel syndrome, and trigger finger. Early detection of amyloidosis is critical for the treatments to be most effective; therefore, screening tissues associated with these conditions for amyloid deposits at the time of corrective surgery may assist physicians with early detection and appropriate clinical management of these patients.
The ACTP is collaborating with physicians in the Departments of Neurosurgery and Orthopedic surgery to obtain biopsy samples from tendons and ligaments obtained following routine surgeries to correct these conditions. We are looking for amyloid deposits in these tissues using a dye called Congo red, and scoring the amount of amyloid that is present. For those tissues that reveal amyloid deposits, further staining is performed to determine the type of amyloid when feasible.
This is a pilot study with the primary goal of determining the incidence of amyloid detection in biopsies obtained during routine corrective surgery at our institution. We plan to expand the study and incorporate whole body imaging using one of our reagents to assess the presence of amyloid in other organs. We are grateful to the UTMC Volunteer Auxiliary for providing funds for these initial studies.
Assessing risk of developing light chain amyloidosis using patient serum.
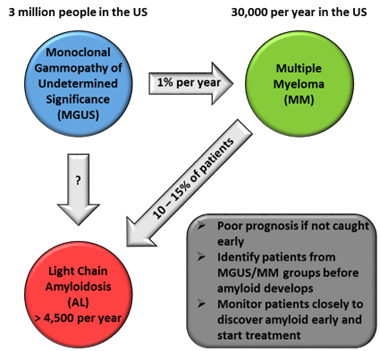
The development of AL amyloidosis involves an underlying plasma cell condition in which abnormal amounts of free antibody light chain proteins are produced. Approximately 3 million people in the United States live with a benign plasma cell condition known as monoclonal gammopathy of undetermined significance (MGUS). In rare instances, and for reasons that remain unknown, MGUS can lead to AL amyloidosis, or a plasma cell cancer known as multiple myeloma (MM), and up to 15% of MM patients may develop symptomatic AL.
We are working on a blood test that may identify which patients from the MGUS or MM populations are at risk of developing amyloidosis. With the help of physicians in the Cancer Institute and the Mayo Clinic in Rochester, we are testing blood samples from these patient groups to use in the development of a potential risk assessment assay.
Translational Research in Therapeutics
The ACTP has experienced researchers with a wide range of technical capabilities. These include molecular biology, protein chemistry, immunohistochemistry, monoclonal antibody production, radiochemistry, small animal PET/CT, SPECT/CT and fluorescence imaging, as well as unique murine amyloidosis models and transgenic mouse strains. These resources have enabled us to develop and characterize novel approaches to the treatment of amyloidosis - focusing principally on methods to remove tissue deposits.
Amyloid Therapeutics: Fc- and Antibody-peptide conjugates. With the aim of creating a pan-amyloid therapeutic reagent, we initially generated, by molecular cloning, a hybrid immunoglobulin Fc-peptide fusion, Fcp5, that combines the broad-spectrum amyloid reactivity of our imaging peptide technology with a partial antibody fragment. This reagent thus allows amyloid targeting in tissues and engagement of immune cells that can take up and clear the deposits.
The success of this approach led us to develop a prototypic antibody-peptide fusion capable of the same amyloid targeting and clearing but using a complete antibody as the cell activator.
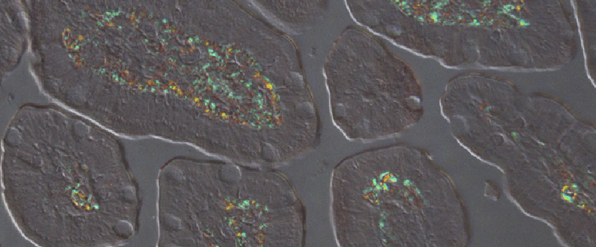
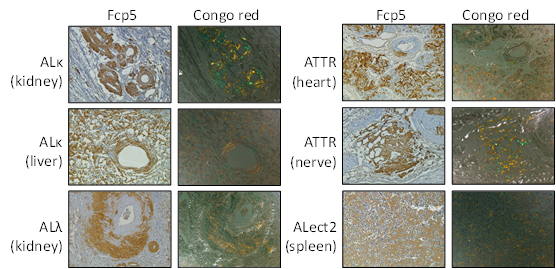
The Fcp5 molecule binds avidly to multiple types of amyloid and in various organs, which we showed using amyloid laden tissue samples.
The Fc- and antibody-peptide fusions have been further adapted in collaboration with industry partners, and the antibody is being assessed in clinical studies.
Amyloid Therapeutics: Chimeric Antigen Receptor Macrophages
Chimeric antigen receptors (CAR) are man-made proteins that can be incorporated into cells to enhance their functionality or expand their recognition of molecules. Macrophages are white blood cells that usually clear misfolded proteins and other foreign entities, such as bacteria, but for reasons we are only recently appreciating, they do not naturally clear amyloid in most patients with amyloidosis. To help promote recognition and clearance of amyloid by these cells, we are developing CAR molecules to incorporate into macrophages, so called CAR macrophages or CARM.
Using our pan-amyloid reactive peptides we have successfully built CARs and introduced them into macrophages.
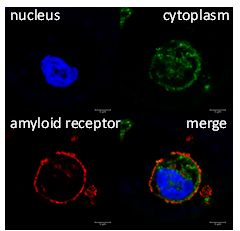
Expression of these engineered, amyloid-reactive CAR proteins on the surface of macrophages enhances recognition of amyloid and enhances phagocytosis (uptake) of the material.
Although still in the early stages of development, we anticipate that an amyloid-reactive CARM may provide a complimentary approach to the goal of tissue amyloid removal when used with other amyloid-clearing immunotherapeutics, such as amyloid reactive antibodies.
Oncology Research
The focus of the cancer research within the Amyloidosis and Cancer Theranostics Program is two-fold. First, we are studying the interaction between the human brain cancer, glioblastoma (GBM) and an oncolytic herpes simplex virus (oHSV). We are engineering oncolytic herpes simplex viruses (oHSV) to combat glioblastoma with the impetus on arming oHSV with immunotherapeutic payloads designed to induce anti-tumor immunity. Through collaborative relationships with the University of Pittsburgh and Harvard Medical School we are evaluating the safety and efficacy of several of these oncolytic vectors clinically.
Recently, we have been awarded a multicenter Program Project Grant through the National Insitute's of Health, National Cancer Institue to study combinatorial immunotheraputic approaches to combat GBM. This work will evaluate whether combining oHSV therapy with immune checkpoint inhibitors (e.g., anti-PD-1) can work synergistically to enhance glioma survival outcomes and induce anti-cancer immunity.
CLIA-Approved Congo Red Histology Testing Facility
The Amyloidosis and Cancer Theranostics Program (ACTP) is a multi-disciplinary, collaborative, translational research program focused on all aspects of systemic amyloidosis. Given its vast experience in the study of amyloid and diagnostic testing associated with detection of the disease, the ACTP has developed significant expertise in preparation and interpretation of tissue staining protocols for the detection of amyloid, a technique that is critically important for the effective clinical care of patients.
The detection of amyloid in clinical samples remains challenging and relies, for the most part, on the staining of tissue biopsy samples with a dye called Congo red. Using a specialized microscope, amyloid deposits appear as green-gold, or red-green birefringent material in the tissue. This remains the ground truth standard for the presence of amyloid and is used to diagnose the vast majority of systemic amyloid types.
In 2020 the ACTP obtained Clinical Laboratory Improvement Amendments (CLIA) certification (44D2185179) for the performance and interpretation of the Congo red staining of patient samples obtained from subjects suspected of having amyloidosis. To perform testing under the Clinical Laboratory Improvement Amendments of 1988, Public Law 100-578, the laboratory complies with all CLIA requirements. These requirements are found in section 353 of the Public Health Service Act (42 U.S.C. § 263a) and 42 Code of Federal Regulations, Part 493 (42 C.F.R. § 493).
The ACTP is now routinely performing Congo red staining of clinical samples and, with the support of the Dept. of Pathology, providing reports that can be used for the diagnosis of the disease in patients seen at UTMC and area hospitals.
Outreach
Amyloidosis Support Group Meetings. The ACTP works closely with members of the Amyloidosis Foundation to conduct support group meetings for patients and their caregivers. We try to hold two meetings annually, and with the help of the Amyloidosis Foundation, we are occasionally able to host amyloidosis experts from various academic medical centers and pharmaceutical companies around the United States. We strive to provide the most current information on the disease, and as researchers, we value patient input into future areas of research. We appreciate that the Cancer Institute offers facility access to hold these meetings. For more information, email Dr. Emily Martin at emartin@utmck.edu.
Without the engagement of all the incredible patients who support and encourage our work, we would not have been able to achieve many of our goals. We are truly grateful for their help.
ACTP Laboratory Resources
- Molecular biology, transfection, and cloning laboratory
- Preclinical animal model laboratory
- Cell culture facilities
- Immunohistochemistry and histology laboratory
- Hybridoma and monoclonal antibody production laboratory
- Protein chemistry and biophysics laboratory
- Cell biology laboratory
- Recombinant protein and peptide phage display laboratories
- Protein-peptide assay development laboratory
- Optical imaging and preclinical assay lab
- Amyloid and tissue sample repository
- Microscopy facility
- Preclinical Imaging Facility
- Flow cytometry laboratory
Partnership with Attralus, Inc.
Attralus is a clinical stage biotechnology company dedicated to the development of therapeutics and diagnostic agents for amyloidosis. Attralus aims to transform the lives of patients with systemic amyloidosis with therapeutics that remove toxic amyloid. Members of the ACTP were involved in founding Attralus, which has licensed technologies from the Program, through the University of Tennessee Research Foundation. Attralus is currently evaluating amyloid-clearing therapeutics in the clinic.
Give to the Amyloidosis and Cancer Theranostics Program
The Amyloidosis and Cancer Theranostics Research Gift Fund provides the UTHSC COM-Knoxville with additional resources to ensure continued excellence in basic and applied research of amyloid-related diseases and cancers. Your support will assist in the purchase of necessary equipment and supplies as well as grants and stipends for deserving students, residents, and/or faculty.
Additional Resources
Amyloidosis Support Groups
Amyloidosis Foundation
Amyloidosis Research Consortium